By Jerry Grillo
Messenger RNA, or mRNA, has been used to immunize millions of people in just the past few years, leading the world out of a pandemic, and allowing researchers to consider other therapeutic targets for these flexible, effective drugs.
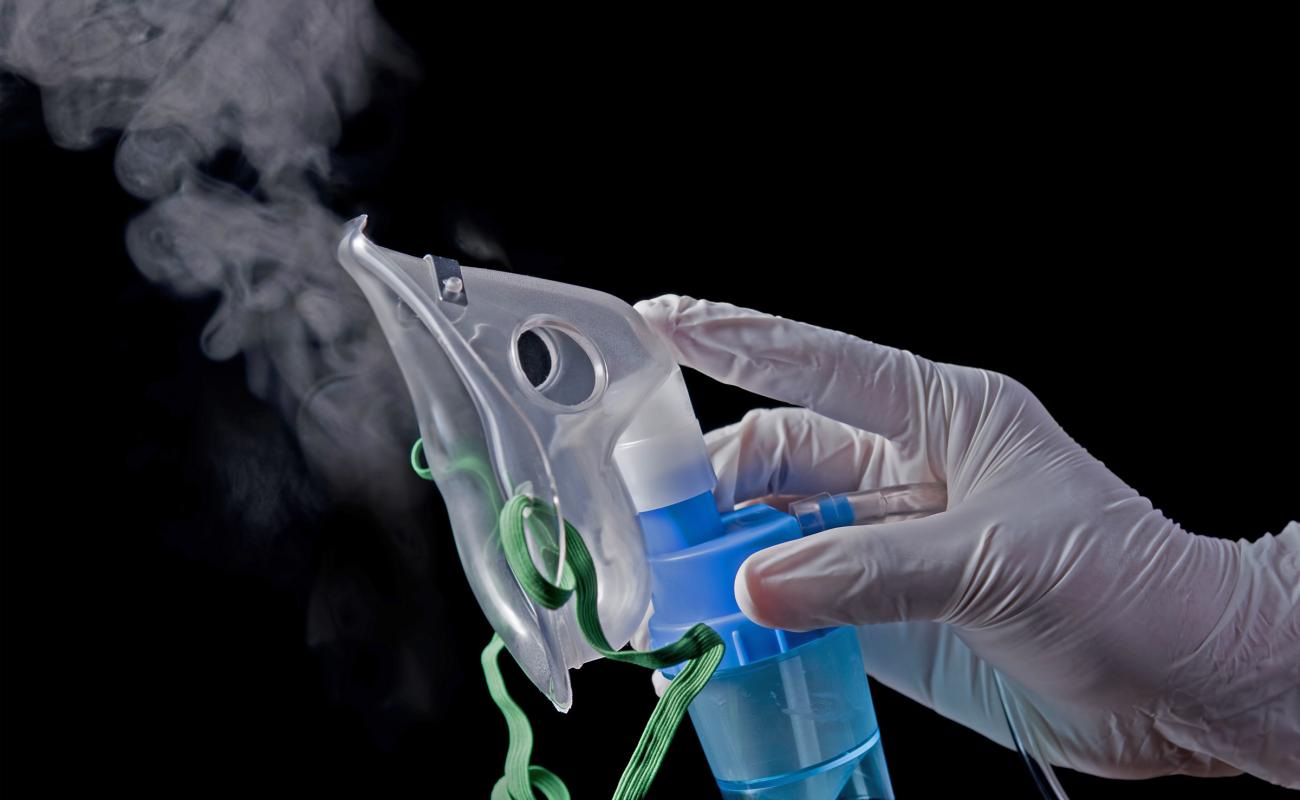
Among the most likely targets for future mRNA therapies are the lungs, given the large number of pulmonary diseases, such as the coronavirus, influenza, asthma, cystic fibrosis, and others.
Now a team of multi-disciplinary investigators from five universities, led by Georgia Tech faculty researchers, has provided a potential path toward that future. In a study published Nov. 28 in the journal Nature materials, they describe polymeric nanoparticle formulations designed specifically for inhalable mRNA delivery, via an easy-to-use nebulizer.
“Nanoparticles made of polymers have specific strengths, and the lung happens to be a place where they are very good for delivery,” said Phil Santangelo, professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
In previous work, Santangelo’s lab also has developed mRNA payloads transported by lipid nanoparticles. But polymeric nanoparticles which are designed to carry drugs to their destinations have a higher loading capacity and are compatible with many compounds.
“In the lung, they’re just flat out better,” Santangelo added. “And we can use them with a wider range of nebulizers. That may not be as important for testing in rodents, but it is when you’re talking about getting this into a person.”
Delivering the Goods
Polymers are large molecules comprised of small, repeating molecular building blocks called monomers. For this study, the researchers focused on synthetic, biodegradable polymers called poly(beta-amino-ester)s, or PBAEs. In a ground-breaking study released last year, Santangelo’s team demonstrated the strength of PBAE formulations in delivering mRNA that can express the CRISPR Cas13a protein in lung tissue, where it was effective in stopping SARS-CoV-2 (coronavirus).
In the latest work, the researchers used a process called combinatorial synthesis – a method for preparing large numbers of chemical compounds – to screen 166 different PBAE formulations. One of these, P76, emerged as the best candidate for protein expression, that is, delivering the therapeutic goods efficiently into the lungs of animals, from mice to non-human primates, which makes P76 species agnostic.
Turns out, the polymer is also compatible with a variety and combination of cargos, which is not typically the case with the delivery of RNAs. But P76 has demonstrated its ability to transport disparate RNAs. So, in addition to being species agnostic, the polymer is mostly cargo agnostic, too.
Using P76, the researchers could effectively encapsulate mRNA together with crRNA, which is a guide strand that basically tells the Cas13a protein who it should target. The key to making it all work so well was incorporating organic compounds called thiols into the mix (turning a PBAE into a PBATE), “dramatically increasing the utility of the polymer for any CRISPR-based therapeutic candidate,” the authors of the study wrote.
“With this new polymer, compared to the old one from our previous work, we get much better protein expression,” Santangelo said. “We can actually decrease dosage by a factor of four, or 400%, and have the same therapeutic effectiveness. That is significant. It’s a striking improvement.”
Santangelo’s lab collaborated with two Georgia Tech faculty members from the School of Chemistry and Biochemistry, Professor M.G. Finn and Associate Professor James Gumbart, who also is a member of the School of Physics. The University of Georgia, University of Louisiana-Lafayette, and Mississippi State University were also part of the study, which involved more than 25 authors.
Their work in Nature Materials was published on the heels of another nebulizer-based study from the Santangelo team that was published in the journal Advanced Science. That work detailed the development of a more efficient, inhalable mRNA medicine to prevent respiratory virus infections like the coronavirus.
In both papers, which were supported by the Defense Advanced Research Projects Agency (DARPA), the researchers demonstrated the utility of polymeric formulations for delivering the potent cargo into the lungs.
“With these studies we basically wanted to make people aware of new versions of a class of molecules that have lots of advantages over the old ones,” Santangelo said. “And the reality is, I think, for the nebulization and delivery to the lung, they have big advantages. These polymers make a lot of sense.”
And potentially, a lot of cents, too, with the global mRNA therapeutics market expected to exceed $26 billion by 2028.
CITATION: Laura Rotolo, Daryll Vanover, Nicholas C. Bruno, Hannah E. Peck, Chiara Zurla, Jackelyn Murray, Richard K. Noel, Laura O’Farrell, Mariluz Araínga, Nichole Orr-Burks, Jae Yeon Joo, Lorena C. S. Chaves, Younghun Jung, Jared Beyersdorf, Sanjeev Gumber, Ricardo Guerrero-Ferreira, Santiago Cornejo, Merrilee Thoresen, [PGR1] Alicia K. Olivier, Katie M. Kuo, James C. Gumbart, Amelia R. Woolums, Francois Villinger, Eric R. Lafontaine, Robert J. Hogan, M. G. Finn, and Philip J. Santangelo. “Species-agnostic polymeric formulations for inhalable messenger RNA delivery to the lung.” (Nature Materials, Nov. 28, 2022) doi.org/10.1038/s41563-022-01404-0
COMPETING INTERESTS: Rotolo, Vanover, Bruno, and Santangelo have a provisional patent filing related to this work. Vanover, Zurla, and Santangelo are cofounders of Tether Therapeutics and this study could affect their personal financial statuses. All other authors declare no competing interests.
Latest BME News
Jo honored for his impact on science and mentorship
The department rises to the top in biomedical engineering programs for undergraduate education.
Commercialization program in Coulter BME announces project teams who will receive support to get their research to market.
Courses in the Wallace H. Coulter Department of Biomedical Engineering are being reformatted to incorporate AI and machine learning so students are prepared for a data-driven biotech sector.
Influenced by her mother's journey in engineering, Sriya Surapaneni hopes to inspire other young women in the field.
Coulter BME Professor Earns Tenure, Eyes Future of Innovation in Health and Medicine
The grant will fund the development of cutting-edge technology that could detect colorectal cancer through a simple breath test
The surgical support device landed Coulter BME its 4th consecutive win for the College of Engineering competition.